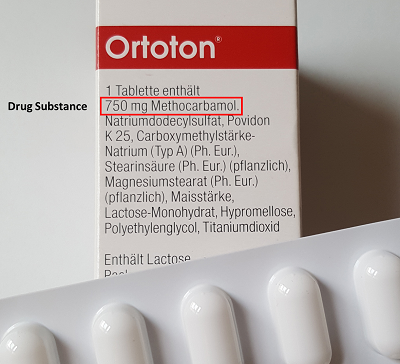
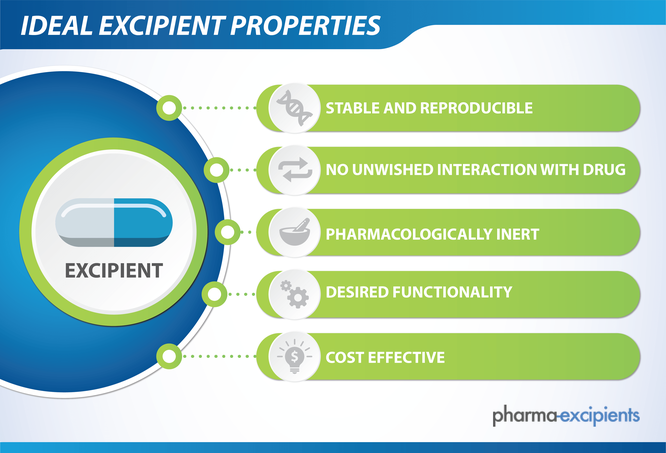
Substances of very high concern under REACH – an evaluation of uncertainties in the environmental risk assessment of endocrine active substances | Umweltbundesamt

GSPR 12. Requirements for devices incorporating active substances or substances absorbed by or locally dispersed in the human body.

Modification of physicochemical characteristics of active pharmaceutical ingredients and application of supersaturatable dosage forms for improving bioavailability of poorly absorbed drugs - ScienceDirect

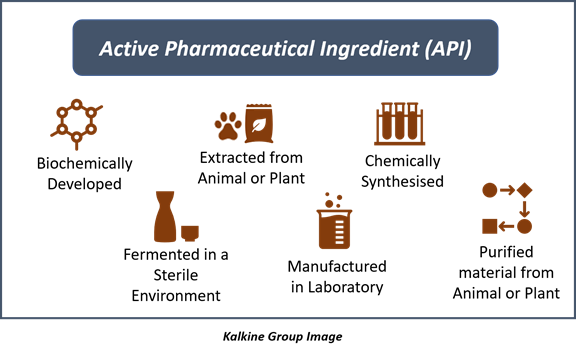




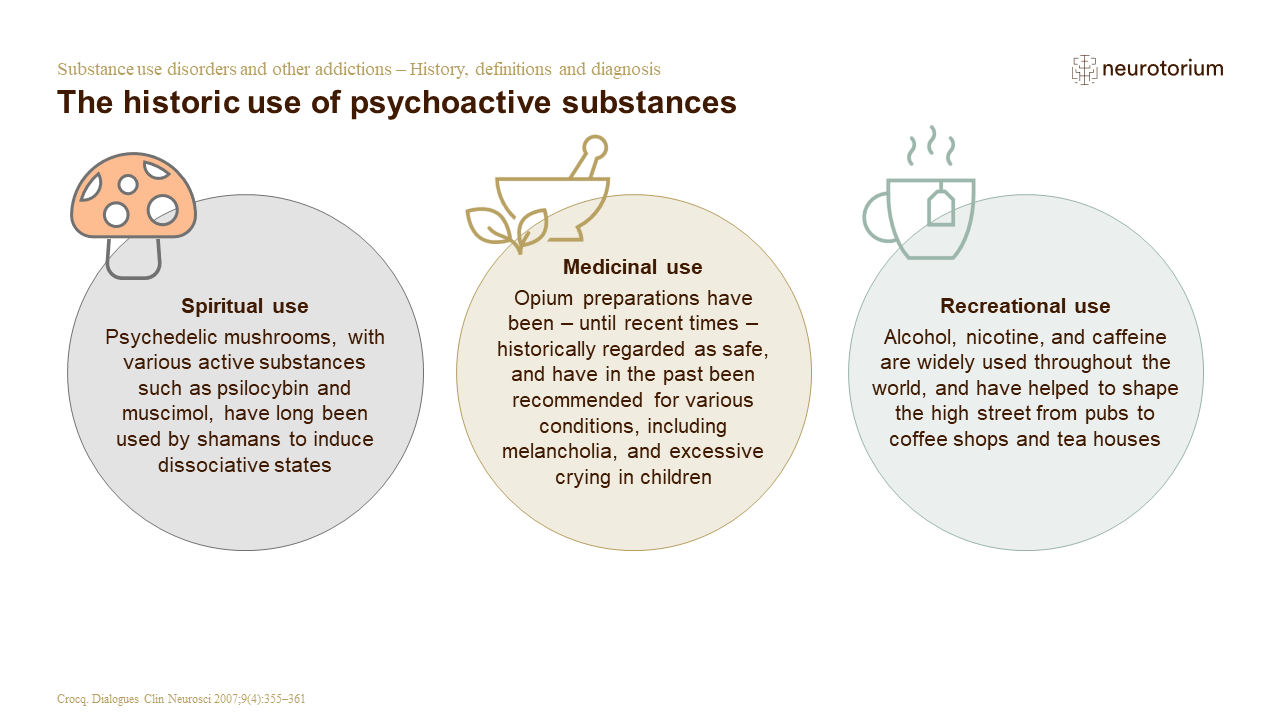

![Pharma] 1.sources of drug & active principles | PPT Pharma] 1.sources of drug & active principles | PPT](https://image.slidesharecdn.com/pharma1-150502144439-conversion-gate01/85/pharma-1sources-of-drug-active-principles-7-320.jpg?cb=1666261283)